 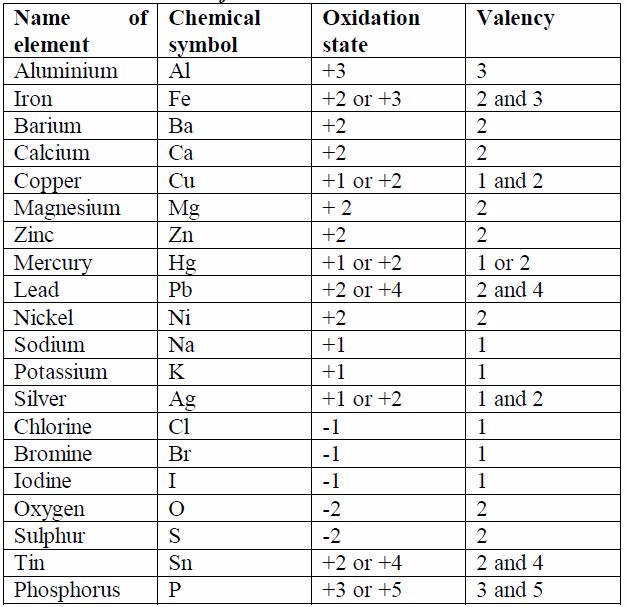
When two or more different elements are chemically joined together, it is known as the compound. When two or more atoms are chemically joined together is known as a molecule. However, there are many things to discuss, but in this article, we will find out the differences between two closely related and confusing terms, which are molecules and compounds. Therefore, we can say atoms play the leading role in chemistry, as it is the smallest unit of matter, but when combined with other atoms may give rise to different elements and compounds. The bond that is shared between two metal atoms is known as a metallic bond. But the ionic or electrovalent bonds are such bonds where the atom donate their electrons to another atom like in NaCl. Secondly, the molecules are determined by the number of atoms taking part in chemical bonding, but compounds are determined by the types of the bond they share between the atoms, which can be either covalent, metallic or ionic bond.Ĭovalent bonds are such type of chemical bonds, in which atoms share the electrons like in O2 (oxygen molecule). are compounds as they are made up of different elements with different atoms. On the other hand, Calcium carbonate (CaCO3), NaCl, H2O, etc. are the molecules, as they have two or more atoms. Let’s take an example to understand in a better way, like Hydrogen gas (H2), Oxygen (O2), water (H2O), HCl, Ozone (O3), etc. So, we can say that any combination of atoms is a molecule, but when the combination of atoms is from different elements, it is known as the compound.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
